 Consider how certain you can be about each advantage and disadvantage.
Consider how certain you can be about each advantage and disadvantage.
It is important to consider the extent to which research provides a good indication of the likely effects of health actions (the certainty of the evidence) when deciding whether to take a health action. For many decisions about health actions, the certainty of the evidence is low or very low.
Explanation
The certainty of the evidence can affect peoples’ choices about health actions. For example, someone might decide not to use or pay for a treatment if the certainty of the evidence is low or very low. How certain the evidence is depends on the fairness of the comparisons, the risk of being misled by the play of chance, how directly relevant the evidence is and the consistency of estimates of effects. Systematic reviews provide the best basis for these judgements and should report an assessment of the certainty of the evidence (based on these judgements).
Treatment recommendations are often based on low certainty evidence and only weak recommendations can be made. Sometimes, usually where nearly everyone would make the same choice despite the uncertainty, it is appropriate to make a strong recommendation despite low-certainty evidence. An example of when a strong recommendation might be made despite low certainty evidence is when there is a life-threatening or catastrophic situation and low-certainty evidence suggests benefit. However, as illustrated by the hydroxychloroquine example below, such decisions can sometimes be deadly.
Even when there is high-certainty evidence, there is almost always some uncertainty about who will benefit, who will not, and who will be harmed. In addition, people vary in how important they consider the benefits and harms of a health action, so decisions may be difficult even when there is high certainty about the effects of a health action.
Uncertainty of the effects of health actions is often inadequately reported or not mentioned in news reports and caution should be used when viewing media reports of studies of health actions.
When there is a public health emergency, it may be appropriate to persuade people to change their behaviour – for example, to wear facemasks – despite important uncertainties about the potential benefits and harms. However, when there are important uncertainties, they should be acknowledged. Not disclosing uncertainties confuses what is known, inhibits research to reduce important uncertainties, and can undermine public trust in health authorities.
Example
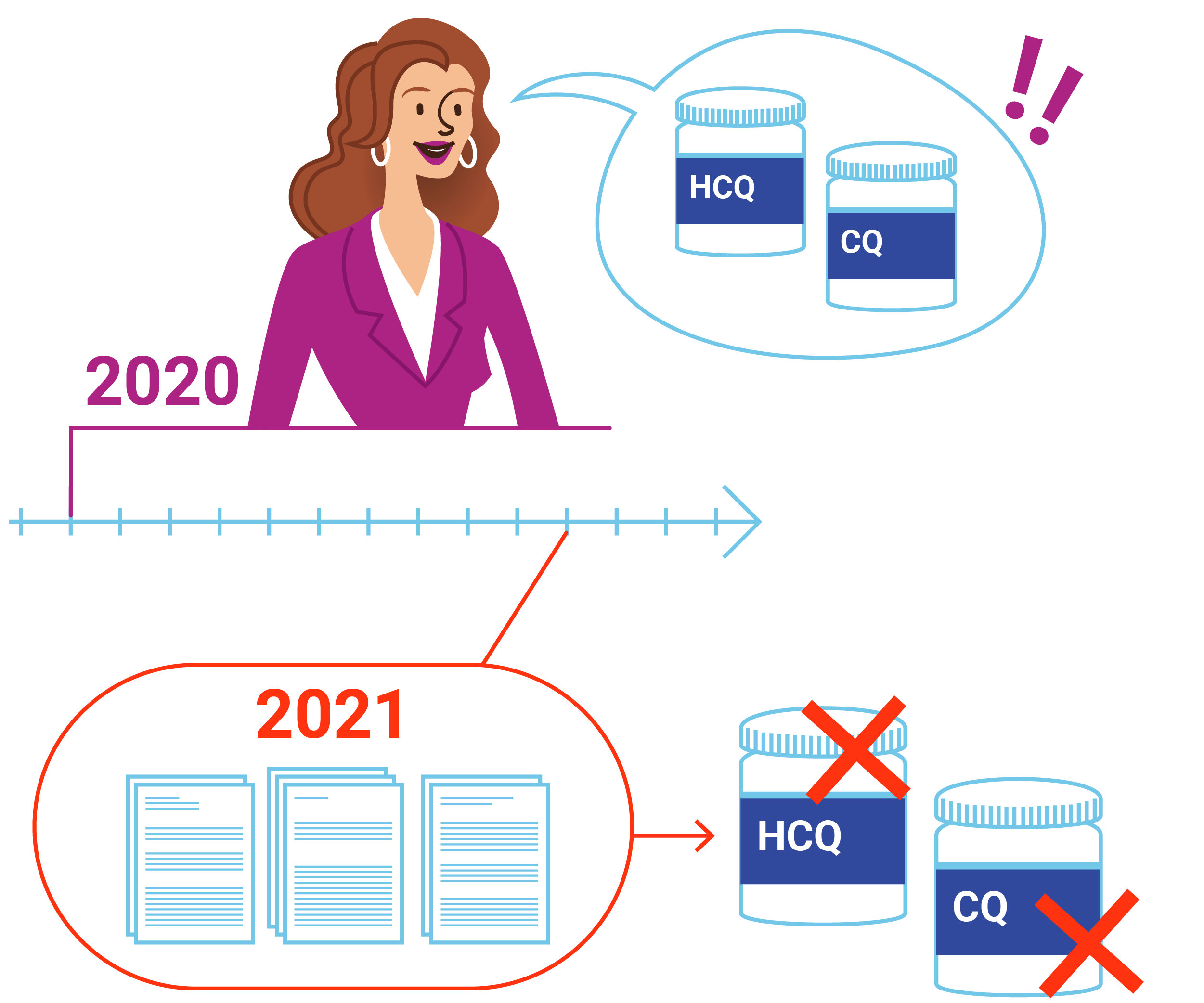
The use of hydroxychloroquine (HCQ) and chloroquine (CQ) to treat Covid-19 illustrates the importance of considering the certainty of the evidence when making decisions about treatments. On March 28, 2020, the US drug licensing authority, the Food and Drug Administration (FDA), issued a letter granting an Emergency Use Authorization for use of HCQ and CQ for treating Covid-19, and the use of HCQ and CQ increased rapidly. The letter did not describe the evidence underlying the decision. It stated that the authorisation was supported by recommendations “for treatment of hospitalized COVID-19 patients in several countries, and a number of national guidelines” based on “limited in-vitro and anecdotal clinical data in case series”. By June, controlled trials had shown that the FDA guidelines had been misleading – no beneficial effects on morbidity or mortality had been detected. On June 15, the FDA revoked the Emergency Use Authorization. A systematic review published in April 2021 found that HCQ increased deaths in Covid-19 patients, and no benefit of chloroquine had been demonstrated.
Remember: Consider the certainty of the evidence when deciding whether to take a health action.
- Video: Dealing with Uncertainty This is a European Food Safety Authority (EFSA) video lasting 3:53 minutes.
- Blog: How certain is the evidence? Students 4 Best Evidence.
